sales@jufuwater.com +8618001362005

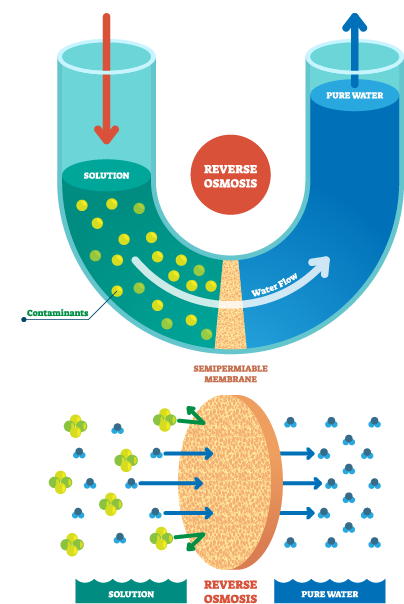
How does Reverse Osmosis work?
When the same volume of dilute solution and concentrated solution are placed on both sides of a container and separated by a semipermeable membrane, the solvent in the dilute solution will naturally pass through the semipermeable membrane to flow to the concentrated solution side, forming an osmotic pressure. If a pressure greater than the osmotic pressure is applied to the concentrated solution side, the solvent will flow to the dilute solution side. This process is called reverse osmosis. The pore size of the reverse osmosis membrane is very small, usually one millionth of a hair (0.0001 micron). Only water molecules and some mineral ions can pass through, while other impurities and heavy metals are intercepted and discharged.